The carbon dioxide reduction reaction (CO2RR) in aqueous media is an important process. The transformation of CO2 into, e.g., fuels could help to alleviate the negative effects of this greenhouse gas. The identification of suitable catalysts that are both efficient and selective for the generation of the desired products is an interesting research target. Multiple useful compounds can be potentially produced via the reduction of carbon dioxide, e.g., carbon monoxide, formate, formaldehyde, or methanol. The competition of the hydrogen evolution reaction (HER) in the presence of water makes the process more challenging.
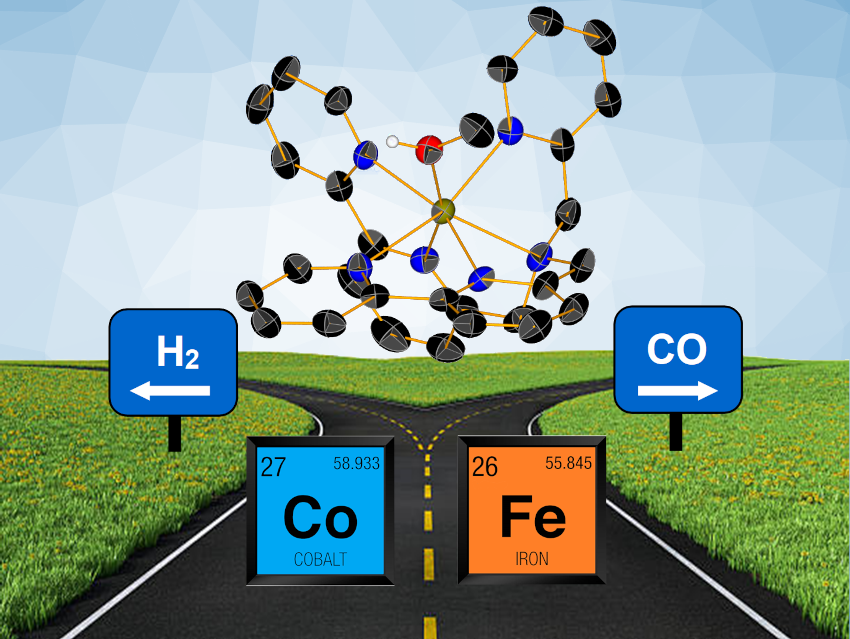
Mariachiara Pastore, University of Lorraine and CNRS, Nancy, France, Albert Ruggi, University of Fribourg, Switzerland, Mirco Natali, University of Ferrara, Italy, and colleagues have investigated the catalytic activity of a cobalt complex and its iron analog, both featuring the same redox-active ligand and an unusual heptacoordinated environment, towards the CO2RR in acetonitrile/water mixtures. The team used DBPy-PyA, or(1-([2,2′-bipyridin]-6-yl)-N-([2,2’-bipyridin]-6-ylmethyl)-N-(pyridin-2-ylmethyl)methanamine, as a redox-active ligand.
The researchers found that during electrolysis, the cobalt complex mainly gives formate—or H2 at a higher voltage and water content, while the iron complex always delivers CO as the major product. Under photochemical conditions with [Ru(bpy)3]2+ (bpy = 2,2′-bipyridine)as a sensitizer and N,N-diisopropylethylamine as an electron donor, the cobalt complex leads to H2 formation, while the iron analog, again, generates CO.
According to the team, the different catalytic activity mainly stems from the tendency of the iron complex to react with CO2 rather than with H+. The work could be useful for the rational development of hybrid photochemical systems that target specific transformations.
- Catalytic CO2 Reduction with Heptacoordinated Polypyridine Complexes: Switching the Selectivity via Metal Replacement,
Federico Droghetti, Agnese Amati, Fabien Pascale, Aurélien Crochet, Mariachiara Pastore, Albert Ruggi, Mirco Natali,
ChemSusChem 2023.
https://doi.org/10.1002/cssc.202300737