Luminescent molecules have many applications, e.g., in bioimaging. Organic molecules with large, conjugated π-systems are one class of compounds suitable for these applications. Their properties can be tuned by varying the size and structure of the π-system, allowing researchers to tailor them to the desired application. One important building block for such molecules is pyrene, a polycyclic aromatic hydrocarbon (PAH) containing four fused benzene rings that shows blue fluorescence in its unsubstituted form.
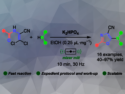
Guangle Niu, Shandong University, Jinan, China, Xing Feng, Guangdong University of Technology and South China University of Technology, Guangzhou, China, and colleagues have developed a one-pot synthesis for pyreno[2,1-b]furans with alkynyl phenyl substituents (example structure pictured). The target molecules were synthesized from 1,3,6,8-tetrabromo-2-hydroxypyrene in a Sonogashira coupling reaction with different phenyl acetylene derivatives, followed by in-situ formation of the furan ring. The reaction was performed with different para substituents at the phenyl rings (OMe, tBu, or CF3), as well as with thiophenyl acetylene.
All four products were isolated as orange powders with low solubility in organic solvents. The structures were confirmed by 1H- and 13C NMR spectroscopy, high-resolution mass spectrometry (HRMS, and—in case of the tert-butyl derivative—by X-ray diffraction. The team performed UV-Vis measurements and found that the four compounds show similar absorption bands between 300–370 nm and 400–525 nm. The fluorescence properties of the four materials are also similar, with intense emissions between 505 and 516 nm. The highest fluorescence quantum yield was observed for the tert-butyl-substituted derivative. All three phenyl-substituted pyreno[2,1b]furans display strong two-photon absorption. These photophysical properties make the new pyrene derivatives promising candidates for applications in bioimaging.
- One-Pot Synthesis of Pyreno[2,1-b]furan Molecules with Two-Photon Absorption Properties,
Xiaohui Wang, Chengjing Zhang, Jin Zeng, Xiaoyu Mao, Carl Redshaw, Guangle Niu, Xiaoqiang Yu, Xing Feng,
J. Org. Chem 2022.
https://doi.org/10.1021/acs.joc.2c01303
![Pyreno[2,1-b]furans Synthesized](https://www.chemistryviews.org/wp-content/uploads/2022/09/pyrenofurans.png)