The cycling of water across membrane transporters is a hallmark of cell metabolism and is potentially of high diagnostic significance for the characterization of tumors and other diseases. Giuseppe Ferrauto, University of Torino, Italy, Silvio Aime, IRCCS SDN SynLab, Napoli, Italy, and colleagues have introduced a new MRI-based method for assessing this water exchange. Using this method, they were able to estimate the degree of malignancy and the success of treatments in mouse tumor models.
Magnetic Resonance Imaging
Not all cancers are equal. Depending on the type of tumor, a given treatment may be spot on or fail completely. For targeted, effective treatment that is as gentle as possible, it is important to precisely locate the tumor and determine its malignancy.
Magnetic resonance imaging (MRI) provides excellent time- and spatially-resolved images for the characterization of tumors. During this procedure, the patient lies in a “tube” in which there is a very strong magnetic field. The spins of protons align themselves in this magnetic field. Radio waves are beamed in and synchronize the precessions of the spins, temporarily flipping some of them. Depending on the composition of the tissue, this “magnetization” is lost at different times (relaxation). This can be used to compute 3D images. Gadolinium contrast agents reduce the relaxation times. These agents are more concentrated in tumors because their blood vessels are particularly permeable. This increases the contrast and makes it easier to define the tumor.
A Biomarker of Cancer “Aggressiveness”
Contrast agents only spread through the extracellular compartments of the tumor; they do not enter tumor cells. The team wanted to exploit this feature to determine the degree of water exchange through the cell membrane. Tumor cells are more metabolically active than healthy cells and have more transport proteins and channels in their cell membranes.
These proteins also allow water to enter and exit the cell, and the degree of water exchange is a measure of the aggressiveness of a tumor. Yet, classic MRI cannot show this.
Chemical Exchange Saturation Transfer
The team decided to work with a new MRI method called CEST (Chemical Exchange Saturation Transfer). There is constant proton exchange between free water and hydrogen-containing groups in biomolecules, such as the amine groups in creatine. The radio frequencies at which a proton can be “magnetized” depend on the chemical environment of that proton, so frequencies are different for protons in free water and those bound to creatine, for example.
With a matching pulse, the creatine-bound protons can be saturated. These protons are exchanged and bind to nearby free water. They keep their “saturated magnetization state” as they do this. If radio waves with the right frequency for free water protons are then pulsed, an increasing number of these protons are already magnetized and cannot absorb the energy (the CEST signal in MR images). Absorption decreases until the proton exchange reaches equilibrium. This makes it possible to draw conclusions about the concentration of creatine and other proton-exchanging molecules in a cell, which can be used for cancer phenotyping.
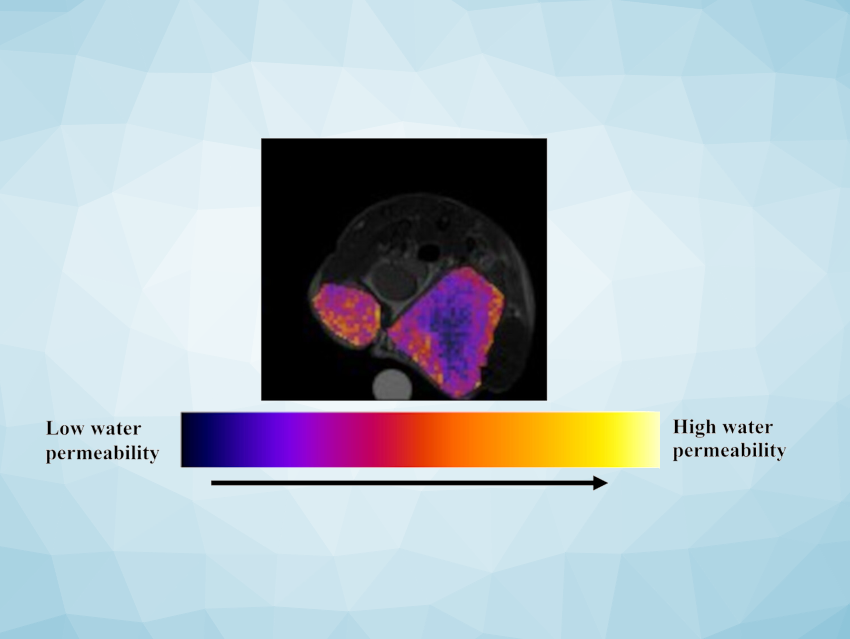
Measuring Water Permeability
If a contrast agent is then administered and enters the extracellular compartment, the magnetization of the water protons there decreases significantly faster. Because water is exchanged through the membrane, the number of magnetized water protons within the cells also decreases more quickly. This, in turn, changes the CEST signals. The changes after addition of contrast agent reflect the permeability of the tumor cell membrane to water.
The team tested this method in mouse models for breast cancer with different degrees of malignancy. As expected, the observable water exchange increases as the tumors grew more aggressive. Within the tumors, it was also possible to differentiate between areas of differing malignancy. The cytostatic drug doxorubicin immediately reduced the water permeability. Hence, the developed method sheds light on tumor phenotypes and provides a new tool to assess the outcome of chemotherapy.
- A Magnetic Resonance Imaging‐Chemical Exchange Saturation Transfer (MRI‐CEST) Method for the Detection of Water Cycling across Cellular Membranes,
Enza Di Gregorio, Chiara Papi, Laura Conti, Antonino Di Lorenzo, Eleonora Cavallari, Marco Salvatore, Carlo Cavaliere, Giuseppe Ferrauto, Silvio Aime,
Angew. Chem. Int. Ed. 2024.
https://doi.org/10.1002/anie.202313485