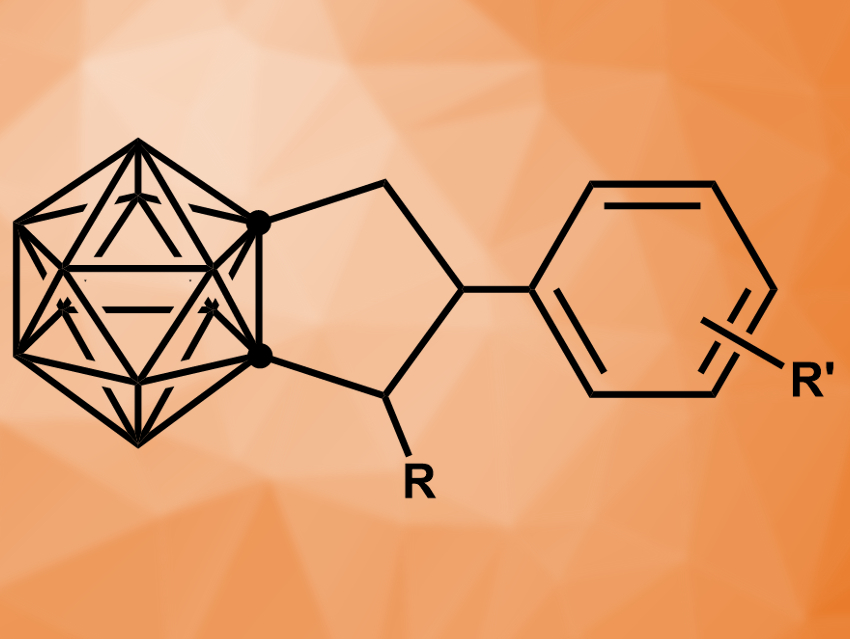
Carboranes are molecular clusters consisting of carbon, boron, and hydrogen. Carborane derivatives can be useful, e.g., as ligands in organometallic chemistry or as boron sources in nuclear medicine. Stable, icosahedral closo-carboranes of the type C2B10H12, called either ortho-, meta-, or para-carborane based on the positions of the carbon atoms, are often used.
Zuowei Xie and colleagues, The Chinese University of Hong Kong, Hong Kong SAR, China, have discovered a magnesium-mediated cyclization that can be used to obtain ortho-carboranes fused to cyclopentane rings (pictured). The team started from 1-arylethynyl-2-alkyl-ortho-carboranes and reacted them with the Grignard reagent MeMgBr. They expected dimethylated products, but instead observed a cyclization involving the two substituents at the carborane.
The team evaluated the substrate scope and found that both electron-donating and electron-withdrawing groups at the para-position of the aryl substituent are tolerated well, with the exception of –F and –CF3. Ortho-substituted systems give lower yields due to steric hindrance. The desired cyclopentane-fused products are formed in moderate to good yields. The researchers propose a mechanism that involves the in-situ formation of HMgBr from the Grignard reagent via nucleophilic substitution of cage B–H bonds. This is followed by the trans-hydromagnesiation of the substituent’s C≡C bond, an intramolecular proton–MgBr exchange, and ring closure.
- Magnesium-mediated sp3 C–H activation in cascade cyclization of 1-arylethynyl-2-alkyl-o-carboranes: efficient synthesis of carborane-fused cyclopentanes,
Jie Zhang, Cen Tang, Zuowei Xie,
Chem. Sci. 2020.
https://doi.org/10.1039/d0sc04465b