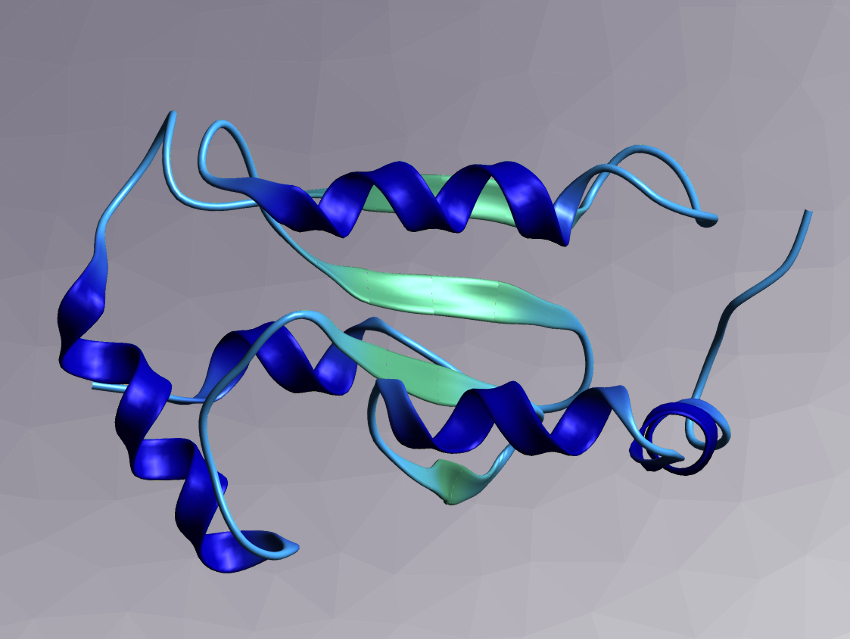
Glutaredoxins (example pictured) are small proteins. Class I glutaredoxins are enzymatically active in redox reactions, for example, in the synthesis of DNA precursors. The substrate of these enzymatically active glutaredoxins is glutathione. There are also class II glutaredoxins, which are enzymatically inactive proteins that can bind iron-sulfur clusters. These proteins are important in the metabolism of iron. Which structural differences are responsible for these different functions had not been fully understood so far.
Holger Gohlke, University of Düsseldorf, Germany, and Forschungszentrum Jülich GmbH, Germany, Bruce Morgan, Saarland University, Saarbrücken, Germany, Marcel Deponte, University of Kaiserslautern, Germany, and colleagues have investigated these structural differences. The team used the yeast glutaredoxin ScGrx7 as a model system and introduced mutations to study the effects of the resulting structural changes.
NMR spectroscopic measurements had shown that at least four protein areas differ between enzymatically active and inactive glutaredoxins. The team identified and characterized key residues from these areas and quantified their contribution to the enzymatic activity by systematically modifying the proteins via genetic mutations. They studied the resulting proteins using enzymatic assays and molecular dynamics (MD) simulations.
The researchers found that all four known protein areas that have structural differences directly or indirectly affect the activity. A loop in the protein structure caused a particularly important difference: It is longer in enzymatically inactive glutaredoxins. When the researchers artificially shortened it, activity increased slightly. Further targeted mutations allowed the team to convert an inactive class II glutaredoxin to an active enzyme. Overall, the work provides an improved understanding of the mechanism of glutaredoxin catalysis.
- Quantitative assessment of the determinant structural differences between redox-active and inactive glutaredoxins,
Linda Liedgens, Jannik Zimmermann, Lucas Wäschenbach, Fabian Geissel, Hugo Laporte, Holger Gohlke, Bruce Morgan, Marcel Deponte,
Nat. Commun. 2020.
https://doi.org/10.1038/s41467-020-15441-3