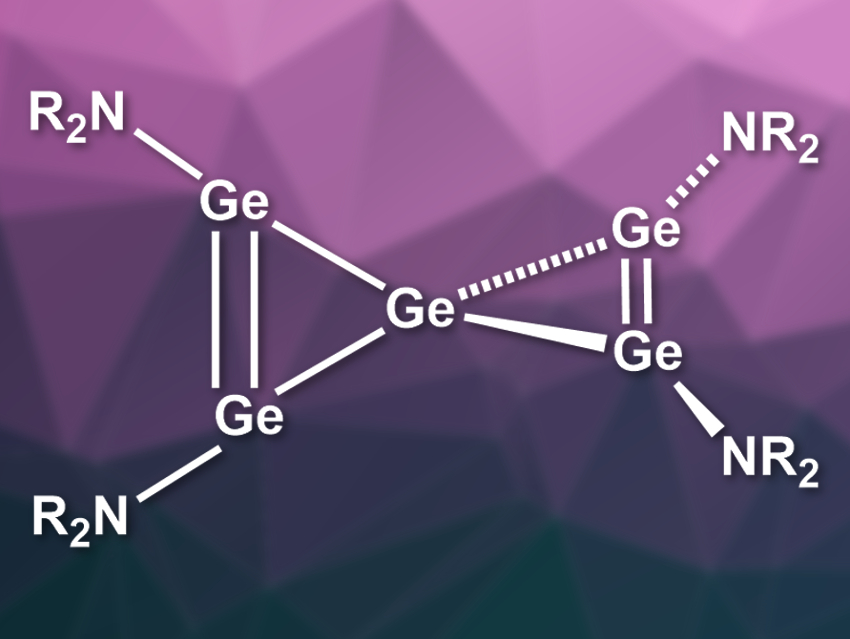
Spiropentadiene is a very strained and thermodynamically unstable organic molecule. The spiropentadiene derivatives that have been synthesized so far decompose at very low temperatures. Heavier analogues, in which the carbon atoms are replaced by, e.g., silicon or germanium, could be more stable due to their different electronic structures. The first Si5-spiropentadiene, or spiropentasiladiene, was isolated in 2000 [1]. Its analogues with Ge, Sn, or Pb had not been reported so far.
Anyang Li, Wenyuan Wang, Northwest University, Xi’an, China, Matthias Driess, Technische Universität Berlin, Germany, and colleagues have synthesized the first Ge5-spiropentadiene (pictured, R = i-Pr3Si). The team started from the potassium amide KNR2, which was reacted with GeCl2·dioxane to give an amido germylene chloride (Cl-Ge-NR2). Reducing this compound with potassium chips in toluene led to the desired product. The spiropentagermadiene was obtained in 58 % yield and formed dark red crystals.
The researchers propose a reaction mechanism that involves the formation of digermyne, followed by cycloaddition to give a cyclotrigermene. This species is reduced to a radical and deaminated and then reacts with another digermyne to give the product. According to the team, the stability of the spirocyclic compound can be attributed to both σ and π delocalization of the electrons, which stabilize the Ge5 structure and reduce the ring strain.
- A Tetra-amido-Protected Ge5-Spiropentadiene,
Yan Guo, Zhengqiang Xia, Jingjing Liu, Jiaxiu Yu, Shenglai Yao, Weiqun Shi, Kongqiu Hu, Sanping Chen, Yaoyu Wang, Anyang Li, Matthias Driess, Wenyuan Wang,
J. Am. Chem. Soc. 2019.
https://doi.org/10.1021/jacs.9b10946
Reference
- [1] A Stable Bicyclic Compound with Two Si=Si Double Bonds,
Takeaki Iwamoto, Makoto Tamura, Chizuko Kabuto, Mitsuo Kira
Science 2000, 290, 504–506.
https://doi.org/10.1126/science.290.5491.504