Carbohydrate-based macrocycles are important in many biological processes. Crown or aza-crown structures with monosaccharides used as chiral building blocks have been intensely studied. Less attention, however, has been paid to such macrocycles with disaccharides or oligosaccharides being a part of the ring.
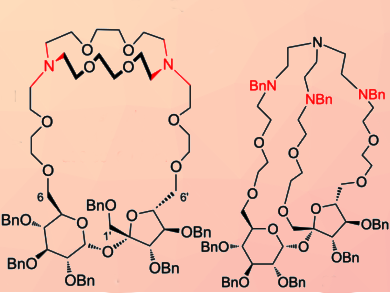
Patrycja Sokołowska, Michał Kowalski, and Sławomir Jarosz, Polish Academy of Sciences, Warsaw, have prepared the first cryptands with a sucrose scaffold. Cryptands with sucrose scaffold were either prepared from readily available 2,3,3’,4,4’-penta-O-benzylsucrose (sucrose triol) or 1’,2,3,3’,4,4’-hexa-O-benzylsucrose (sucrose diol).
Sucrose diol was extended at both terminal positions by five atoms by reaction with bis(2-chloroethyl) ether. Replacement of both terminal chlorine atoms by iodine afforded 1’,2,3,3’,4,4’-hexa-O-benzyl-6,6’-bis[2-(2-iodoethoxy)ethyl]sucrose, which was reacted with a commercially available diaza-crown ether to introduce an additional macrocyclic unit connecting the terminal positions. A sucrose cryptand (pictured right) was afforded in excellent yield (33 %).
The cavity of this cryptand is large. In addition, it is accessible from both sides of the molecule which may decrease the complexing properties, especially of chiral guests. Therefore, the team a second synthesis started with sucrose triol and a tripodal amine to prepare cryptands with cavities of various sizes. For this, they had to elongate all three terminal positions (C1’, C6, C6’) with linkers of different sizes. The researchers reported that the cyclization reaction provided, for example, the cryptand (pictured left) in very high yield: 45.5 % and thus making this reaction a promising strategy to prepare such structures with different sizes.
- First synthesis of cryptands with sucrose scaffold,
Patrycja Sokołowska, Michał Kowalski, Sławomir Jarosz,
Beilstein J. Org. Chem. 2019, 15, 210–217.
https://doi.org/10.3762/bjoc.15.20