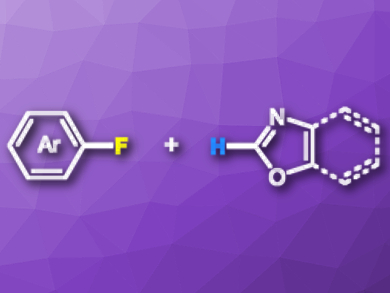
C–F bond functionalization is a useful protocol in the late-stage synthesis of complex organic compounds. However, the formation of C–C bonds via C–F bond cleavage is always challenging because C–F bonds in organic compounds are strong and relatively inert. Therefore, C–F functionalization is usually achieved by using an organometallic compound as a coupling partner. A C–F/C–H cross-coupling can be realized with Pd catalysis or photocatalysis.
Ruopeng Bai, Yu Lan, Chongqing University, Hua Zhang, Nanchang University, both China, and colleagues have developed a coupling reaction of unactivated aryl fluorides and oxazoles using Ni(cod)2 (cod = cyclooctadiene) as a catalyst and 1,3-bis(2,4,6-trimethylphenyl) imidazolium chloride as a ligand. The reactions are carried out in hexane at 160 °C in the presence of tBuONa.
The approach gives the desired arylated oxazoles in moderate to good yields. The synthetic utility of the method was demonstrated by a rapid synthesis of texaline, which is an important natural product with antitubercular activity.
- Ni-Catalyzed C–F Bond Functionalization of Unactivated Aryl Fluorides and Corresponding Coupling with Oxazoles,
Youzhi Yin, Xiaoyu Yue, Qi Zhong, Hanmin Jiang, Ruopeng Bai, Yu Lan, Hua Zhang,
Adv. Synth. Catal. 2018.
https://doi.org/10.1002/adsc.201701506