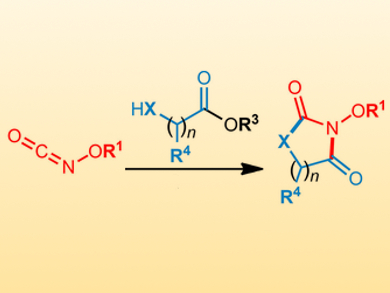
Isocyanates are important reagents in synthetic organic chemistry and have a wide range of applications. While carbon-substituted isocyanates are common, oxygen-substituted isocyanates (pictured) are rare because they are highly reactive and prone to Lossen rearrangements or homotrimerization. However, O-substituted isocyanates could be versatile reagents in the synthesis of bioactive compounds, if their reactivity can be controlled and unwanted reactions can be prevented.
André M. Beauchemin, University of Ottawa, Canada, and colleagues have developed a controlled cascade reaction to synthesize hydantoins and dihydrouracils, using masked O-isocyanates and amino esters as starting materials. The reactions are carried out in MeCN or THF at 70–120 °C. They use i-Pr2NEt or DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) as the base and give the products in good yields.
The researchers have studied the influence of the blocking group on the reactivity and found that phenol is the best one for the cascade reaction because the desired O-isocyanates are conveniently released in situ from the crystalline precursors without causing side reactions. The developed method is the first cascade reaction using O-isocyanates as reactive intermediates.
- Oxygen-Substituted Isocyanates: Blocked (Masked) Isocyanates Enable Controlled Reactivity,
Ryan A. Ivanovich, Dilan E. Polat, André M. Beauchemin,
Adv. Synth. Catal. 2017.
DOI: 10.1002/adsc.201701046