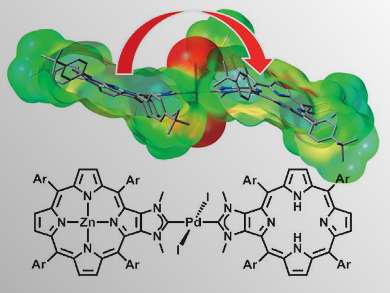
Light energy absorbed by a chromophore, for example chlorophyll or bacteriochlorophyll antennas, is transported to the reaction center through singlet energy transfer and excitation energy migration. In synthetic chromophore analogues, chlorophylls and bacteriochlorophylls are often replaced by zinc(II) porphyrin with rigid spacers, such as anchoring bridges of square-planar trans-PdII and PtII complexes. Nature, however, does not use such heavy metals in the photosynthetic machinery, owing to their cytotoxicity.
Sébastien Richeter, Institut Charles Gerhardt de Montpellier, France, togehter with Romain Ruppert, Université de Strasbourg, France, as well as Pierre Harvey, Université de Sherbrooke, Canada, and colleagues revealed an additional reason why heavy metals are not found in photosystems in nature. The team found that a dyad consisting of a zinc(II) porphyrin and the corresponding free base, [Zn-Fb], fused to N-heterocyclic carbene (NHC) ligands and a bridging trans-PdI2 unit showed unexpectedly slow singlet energy transfer rates despite having favorable structural and spectroscopic parameters. This observation was rationalized by the lack of large molecular orbital (MO) overlaps between the frontier MOs of the donor ([Zn-Fb]) and acceptor (NHCs), which prevented double electron exchange through the trans-PdI2 bridge. Additionally, electronic shielding induced by the linker prevented the two chromophores from fully interacting via their transition dipoles.
These findings suggest that heavy metals are not found in natural photosystems not only for toxicity reasons, but also for the sake of the efficiency of the antenna effect.
- Unexpected Drastic Decrease in the Excited-State Electronic Communication Between Porphyrin Chromophores Covalently Linked by a Palladium(II) Bridge,
Mohammed Abdelhameed, Paul-Ludovic Karsenti, Adam Langlois, Jean-François Lefebvre, Sébastien Richeter, Romain Ruppert, Pierre D. Harvey,
Chem. Eur. J. 2014.
DOI: 10.1002/chem.201403146