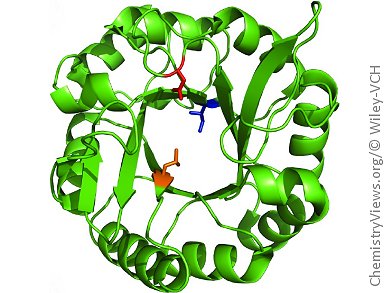
Metalloenzymes are known for catalyzing a wide variety of reactions in nature. Synthetic biologists are hoping to repurpose non-metallic proteins by using metal-coordinating amino acids to make artificial metalloenzymes.
This technique, developed by Jared C. Lewis and colleagues, University of Chicago, IL, USA, uses a well-established method of suppressing natural stop codons with a non-canonical amino acid to incorporate the metal-binding site at any site within the synthetic protein. Once incorporated, the metal-binding amino acid (p-azido-L-phenylalanine; Az) can coordinate rhodium ions, creating the first rhodium metalloenzyme.
In addition to creating a dirhodium-containing ArM, the team demonstrated the ability of this enzyme to catalyze a cyclopropanation reaction and to insert Si–H into a carbon scaffold. Although these are just model reactions, they show the potential for these ArMs to become versatile catalysts for a wide variety of important reactions. The design of the ArMs also allows for modular substitution with other metal-coordinating groups, making them flexible for many different applications.
- A General Method for Artificial Metalloenzyme Formation through Strain-Promoted Azide–Alkyne Cycloaddition,
H. Yang, P. Srivastava, C. Zheng, J. C. Lewis,
ChemBioChem 2014.
DOI: 10.1002/cbic.201300661