Chemical Asymmetry
Chirality, or handedness, is an ongoing topic of discussion for chemists. Living things display handedness, from the spiraling of plant stems and snail shells right down to the double helix of DNA and the twists and turns of the proteins it encodes. For the organic chemist who wants to make compounds that interact with living organisms, such as agrochemicals or pharmaceuticals, chirality can be critical to activity and efficacy—whether they are targeting a crop pest species with an insecticide or attempting to cure a disease.
Common chemical knowledge has it that reactions which give only one chiral form when two are possible require chiral reagents, chiral catalysts, or at the very least chiral auxiliaries that can direct the scheme along the chosen path. On the other hand, magnetism, polarized light, and other physical phenomena have been investigated with the aim to taking control of enantiomeric excess without the need for designer reagents that fit hand in glove, as it were. There were claims for magnetism alone as a chiral agent back in the 1990s, but these were quickly shown to be invalid at the time [1]. While magnetism alone does not have an effect, when it is used in conjunction with incident polarized light, there is some evidence of a usable effect, magnetochiral anisotropy, that can nudge a reaction towards one enantiomer and not the other.
Propagating Chirality
David H. Waldeck, University of Pittsburgh, PA, USA, Yossi Paltiel, The Hebrew University of Jerusalem, Israel, Ron Naaman, Weizmann Institute of Science, Rehovot, Israel, and colleagues have turned the concept of controlling chirality in a reaction on its head. They looked at whether electron spin can be used as a chiral “reagent” to direct a chemical reaction. Electron spin is a quantum phenomenon, the intrinsic angular momentum of the electron. It can be “up” or “down” (or arbitrarily “left” and “right”, if you like). Because its value is 1/2, one might picture it as being akin to a Moebius strip where one has to make two complete rotations, or circuits, to get back to the starting point, and the circuit can be clockwise or counterclockwise.
The team suggests that electrons can be injected into a reaction from a magnetized electrode. Here they can be adsorbed on to a molecule with a distinct spin orientation relative to their velocity. It is, the team asserts, then possible to replace a conventional enantiopure chemical reagent by such spin-polarized electrons and provide the chiral bias for enantioselective reactions. The researchers have demonstrated this concept using three separate proof-of-principle experiments.
“The electron spin itself cannot have a chiral specific interaction. However, when the electron is in motion, its linear momentum and spin define helicity,” Naaman explains. “One can view the electron as a bullet rotating to the right (positive helicity) or to the left (negative helicity) relative to its propagation. Spin with specific helicity can interact with a chiral molecule with a specific handedness,” he adds.
Selection Process
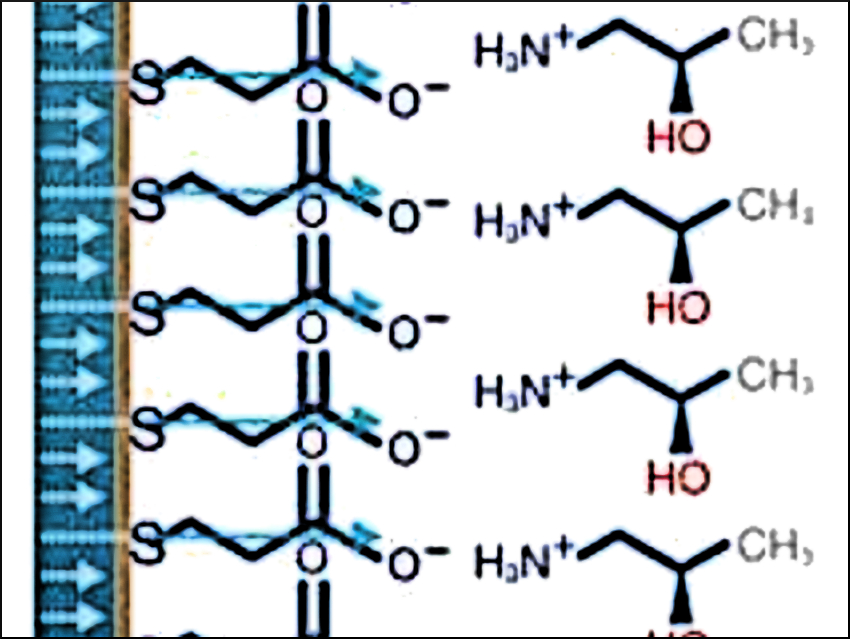
In their first experiment, the team demonstrates the enantioselective association of a chiral molecule, (S)- or (R)-1-amino-2-propanol (pictured right), with a self-assembled monolayer film of achiral carboxyl-terminated alkanethiols, which is spin-polarized on a magnetic material (pictured left). In the second and third experiments, the chiral bias induced by the electron helicity drives enantiospecific electrochemical reduction or oxidation reactions, e.g., involving chiral ferrocene derivatives or camphorsulfonic acid.
The team points out that it is not an enantiospecific interaction of the molecule with the ferromagnetic electrode that induces the chiral bias in the system, but the polarized spin that crosses the interface between the substrate and the molecule. “In all three cases, the direction of the electron spin polarization defines the sense (left-handed versus right-handed) of the enantioselectivity,” the team concludes. They say, their work, thus, demonstrates an entirely novel mechanism for carrying out enantioselective reactions without the need for chiral reagents or catalysts.
- The electron spin as a chiral reagent,
Ron Naaman, Tzuriel S. Metzger, Suryakant Mishra, Brian P. Bloom, Naama Goren, Avner Neubauer, Guy Shmul, Jimeng Wei, Shira Yochelis, Francesco Tassinari, Claudio Fontanesi, David H. Waldeck, Yossi Paltiel,
Angew. Chem. Int. Ed. 2019.
https://doi.org/10.1002/anie.201911400 - [1] D. Clery, D. Bradley, Chemistry. Underhanded ‘breakthrough’ revealed, Science 1994, 265(5168), 21. https://doi.org/10.1126/science.8016651
Also of Interest
- Clever Picture: What is Chirality?, ChemViews Mag. 2017. https://doi.org/10.1002/chemv.201700044
Right- and left-handed structures occur almost everywhere