Thermal Decomposition Examined
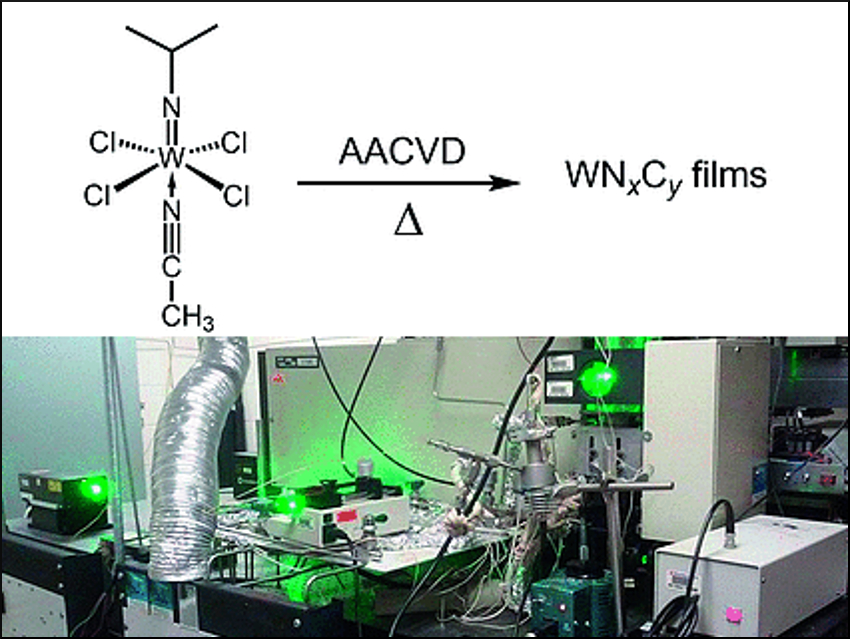
Thin films play a key role in the production of electronics. They can be grown directly on a substrate surface through the chemical vapor deposition (CVD) process, which involves a reaction of vapor phase precursor compounds. Based on in situ Raman spectroscopy during simulated CVD in a customized reactor, the decomposition of a tungsten carbonitride precursor was examined under realistic conditions. Lisa McElwee‐White, University of Florida, Gainesville, USA, and colleagues have proposed a decomposition mechanism.
One of the major advantages of CVD over physical techniques is its conformal film growth, which enables uniform coverage of complex three-dimensional surfaces, including extremely fine structures on wafers. Such conformal film growth is needed for the preparation of diffusion barriers for copper-metalized integrated circuits. Copper atoms from conducting tracks tend to diffuse into the surrounding silicon or silica, altering the electrical properties and eventually causing the failure of the microelectronic components. While current diffusion barriers consist of tantalum/tantalum nitride bilayers applied through physical vapor deposition, alternative materials applied by CVD have been considered. Tungsten carbonitride (WNxCy) is a promising candidate due to its low resistivity, suitable thermal and mechanical stability, and minimal chemical reactivity with other materials used in integrated circuits.
Tungsten Carbonitride Precursor
To establish a suitable CVD process, the nature of the precursor compound is essential. Its physical and chemical characteristics and the mechanisms of its decomposition pathways are critical for controlling the deposition and the properties of the deposited material. “Unfortunately, the vast majority of decomposition characterization is done using techniques that do not capture CVD conditions”, says Lisa McElwee-White. Working with her team, she was able to overcome these limitations by simulating the CVD process in a special, custom-made reactor. This reactor is equipped with a Raman spectrometer, allowing for the observation of gas-phase reaction products in situ. Raman spectroscopy relies on alterations of the vibrational and rotational modes of molecules. As a precursor compound, the researchers chose the tungsten imido complex Cl4(CH3CN)WNiPr, a known precursor for the aerosol-assisted (AA)CVD of tungsten carbonitride thin films.
Based on the observed intermediates combined with the results of previous computational and ex-situ analytical data, the researchers were able to propose a probable decomposition mechanism for the precursor they studied. It includes a reaction known as σ-bond metathesis between the precursor W–Cl bond and H2. This reaction is usually unfavorable in conventional organometallic reaction conditions. “That this reaction occurs might be due to the high temperatures and special conditions inside our CVD reactor”, says McElwee-White. Another step is homolysis of the precursor’s imido bond at the Nimido-C position. This step is also a high-energy reaction that requires high temperatures. “Our findings might explain the limiting deposition temperature observed for the growth of WNxCy from imido complexes.”
- In Situ Investigation of the Thermal Decomposition of Cl4(CH3CN)W(NiPr) During Simulated Chemical Vapor Deposition,
Michelle M. Nolan, Seo Young Kim, Arijit Koley, Tim Anderson, Lisa McElwee-White,
Eur. J. Inorg. Chem. 2019.
https://doi.org/10.1002/ejic.201900627