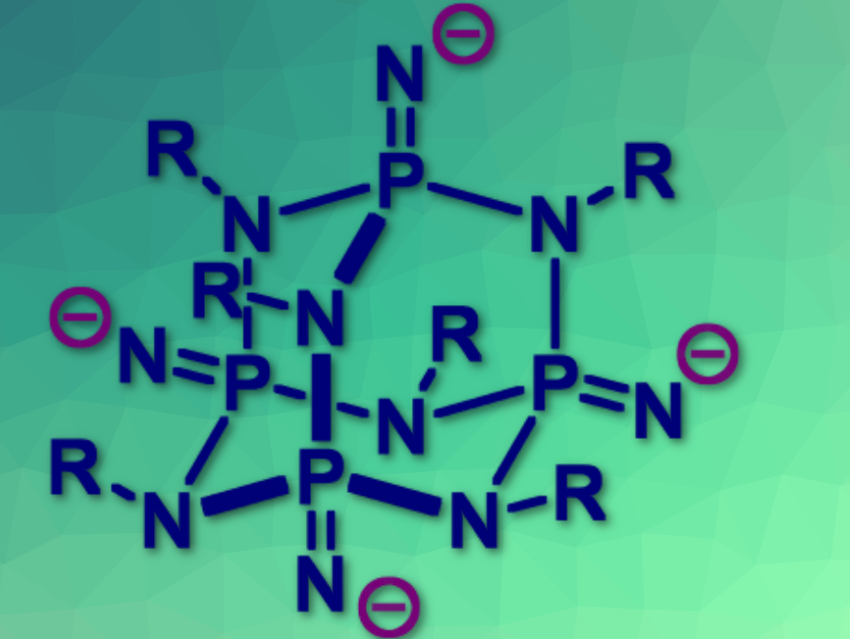
Rigid tetrahedral compounds are valued as molecular scaffolds upon which further covalent chemistry can be carried out. Historically, chemists have used only two such compound classes, tetraarylmethanes and adamantanes. However, they are difficult to synthesize and modify.
Saurabh Chitnis, Dalhousie University, Halifax, Canada, and colleagues have achieved four-fold oxidation of a phosphorus–nitrogen cage (1) using trimethylsilyl azide to access an air-stable tetra-silylphosphinimine cage (PNSiMe3)4(NMe)6 (2) in a multigram scale synthesis.
Stability tests showed that 2 is robust against heat, nucleophiles, oxidants, reductants, electrochemical extremes, and UV light. However, while its core is inert, the surface of 2 has nucleophilic Si–N bonds that react with halides, allowing tetrahedral extension to form polymers and networks.
Derivatives of 2 can be studied by 31P nuclear magnetic resonance (NMR) spectroscopy and they feature lone pairs for coordination chemistry. While tetraarylmethanes and adamantanes enforce a separation between connected groups of 6.9 Å and 2.50 Å, respectively, compound 2 offers a complementary intermediate separation of 5.9 Å.
- (PNSiMe3)4(NMe)6: A Robust Tetravalent Phosphaza‐adamantane Scaffold for Molecular and Macromolecular Construction,
Joseph Bedard, Nicholas Roberts, Mohsen Shayan, Karlee L Bamford, Ulrike Werner-Zwanziger, Katherine M. Marczenko, Saurabh S. Chitnis,
Angewandte Chemie International Edition 2022.
https://doi.org/10.1002/anie.202204851
.PNG)



![Synthesis of [c2]Daisy Chains via Mechanochemistry](https://www.chemistryviews.org/wp-content/uploads/2025/04/202504_RotaxanesWithSolidStateMechanochemistry-125x94.png)