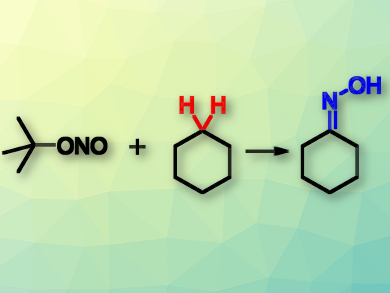
Cyclohexanone oxime (pictured right) is an important precursor for the preparation of ε-caprolactam, which is then used in the production of nylon on a scale of millions of tons per year. The oxime is usually produced by converting cyclohexanone, but there are photoinduced approaches which directly transform cyclohexane and, thus, save reaction steps. These methods, however, use either corrosive gases or energy-inefficient mercury vapor lamps.
Thomas Schaub, CaRLa—Catalysis Research Laboratory, Heidelberg, and BASF SE, Ludwigshafen, both Germany, and colleagues have modified this process to use efficient high-power UV light-emitting diodes (LEDs). The team mixed t-butyl nitrite, cyclohexane, and t-butyl alcohol as an additive, and irradiated the mixture using LEDs at 405 nm and 50 °C for 16 h.
Cyclohexanone oxime was isolated in a combined yield of 76 %. The reaction proceeds via a photonitrosation of cyclohexane, followed by a tautomerization to give the final product. The LEDs are highly energy-efficient and relatively inexpensive. According to the researchers, this method could be a starting point for developing an alternative to the current industrial processes for the synthesis of ε-caprolactam.
- Photoinduced Direct Conversion of Cyclohexane into Cyclohexanone Oxime using LEDs,
Jędrzej Wysocki, Joaquim Henrique Teles, Richard Dehn, Oliver Trapp, Bernd Schäfer, Thomas Schaub,
ChemPhotoChem 2017.
DOI: 10.1002/cptc.201700151