Dr. Ryan Steed, University of North Carolina Asheville, USA, discusses their recently published study investigating whether certain quinoline-based compounds can block the energy system of important antibiotic-resistant pathogens. Some compounds showed strong antibacterial activity, supporting that ATP synthase could be a promising target for new antibiotics.
ATP synthase is the enzyme responsible for producing ATP (adenosine triphosphate), the molecule that carries energy within cells.
What did you do?
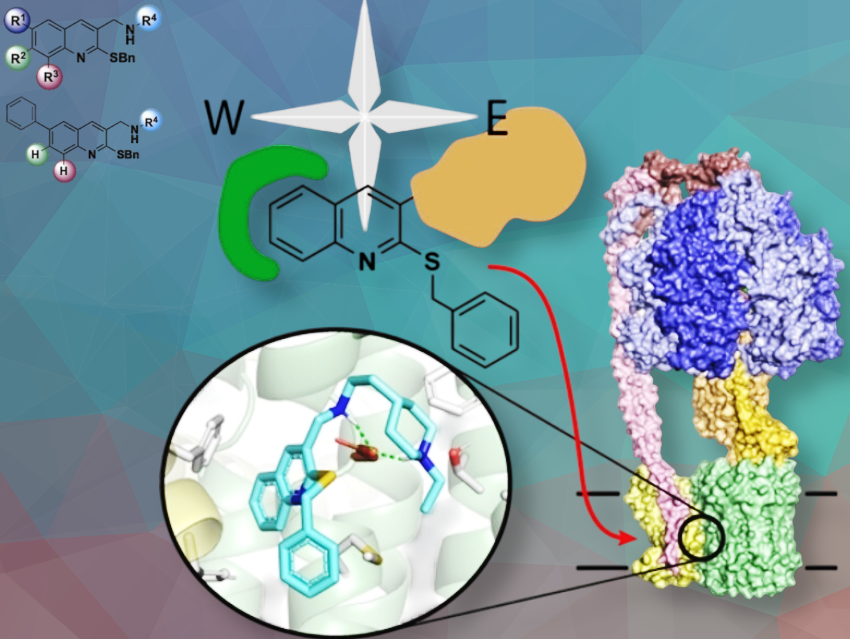
We synthesized new molecules, built on a quinoline scaffold, that inhibit ATP synthesis activity and growth of two pathogenic bacteria, Acinetobacter baumannii (AB) and Pseudomonas aeruginosa (PA), which are both problematic organisms in healthcare settings.
AB is a highly drug-resistant bacterium often associated with hospital-acquired infections, particularly in immunocompromised patients. PA is an opportunistic pathogen known for causing infections in individuals with weakened immune systems, including those with cystic fibrosis or burn wounds.
Why are you doing this?
Antibiotic resistance is a growing problem worldwide, and a continuous pipeline of new antibiotics is needed, especially compounds with novel mechanisms, to challenge pathogenic bacteria in new ways.
Most antibiotics target a handful of cellular processes like protein synthesis, DNA synthesis, and cell wall construction. We are looking to find compounds that target energy production in bacteria, specifically oxidative phosphorylation.
Bacterial bioenergetics is an especially promising target since it is necessary for the survival of many pathogenic bacteria, and it has been underexplored so far, so bacteria have fewer pre-existing defenses against compounds targeting these processes.
What is new and cool about your work?
This is the first report of compounds that inhibit Acinetobacter baumannii ATP synthase and inhibit the growth of clinically-derived, drug-resistant AB. This result shows that ATP synthase is a viable target for the development of new antibiotics against this problematic bacterium.
What is the main significance of your results?
We began using the quinoline scaffold several years ago to make compounds targeting ATP synthesis in Pseudomonas aeruginosa. Previously, we had found that bulky amine substituents on one side of the molecule (referred to as the “east” side) increased potency against PA.
In the current study, we show that adding an additional ring on the “west” side of the quinoline increases potency against PA and now Acinetobacter. Additionally, we observed differences in activity against PA vs. AB, which is a step toward designing compounds with molecular selectivity between two closely related enzymes.
What is the longer-term vision for your research?
Our results are promising, in that ATP synthase is a workable target for antibiotics, but these molecules are still in the early stages of development.
We intend to further improve the molecular selectivity so that we can specifically target the ATP synthase of one bacterial species while sparing other, even closely related, enzymes, which should help minimize the toxicity and side effects of any new antibiotics that are derived from our library.
What part of your work was the most challenging?
The work described in this paper was done entirely by undergraduate students, which is a wonderful opportunity to train future scientists in medicinal chemistry, biochemistry, and antibiotic development, but it also comes with challenges. We are more limited than other research labs in the experiments we are equipped to accomplish. Also, it is always an interesting exercise to assemble results from several distinct but related undergraduate research projects into a cohesive work that has an impact on the scientific community.
Thank you very much for sharing these insights.
The paper they talked about:
- Quinoline ATP Synthase Inhibitors with Activity Against Multidrug Resistant Acinetobacter baumannii and Pseudomonas aeruginosa,
Katie T. Ward, Alexander P. L. Williams, Angelina L. Dennison, Lena Aamir, Darien L. Allen, Britza Chavez-Arellano, Toni A. Marchlewski, Mars L. Zappia, Amanda L. Wolfe, P. Ryan Steed,
ChemMedChem 2025.
https://doi.org/10.1002/cmdc.202400952
Ryan Steed is an Associate Professor at the Chemistry and Biochemistry at the University of North Carolina Asheville in Asheville, NC,USA.