Synthetic peptides are promising for the selective delivery of cytotoxic agents to tumor cells without harming healthy tissue. However, the targeting properties of monomeric peptides are not always sufficient to obtain tumor selectivity.
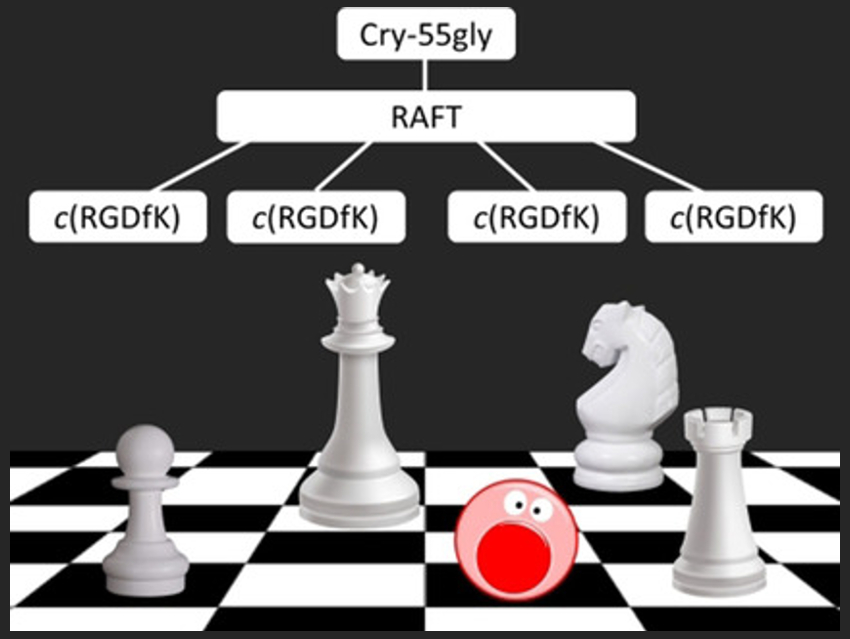
Norbert Sewald, Bielefeld University, Germany, Didier Boturyn, University Grenoble Alpes, France, and colleagues have developed anticancer conjugates (6, pictured below) that contain a multimeric ligand. This system contains a so-called RAFT (regioselectively addressable functionalized template) cyclodecapeptide scaffold and four copies of the cyclic Arg‐Gly‐Asp peptide c(RGDfK) (pictured in blue). This peptide targets the cancer-specific integrin αvβ3 present on the cell surface. Integrins are a type of receptor protein. The multivalent peptides bind better to the integrin than monomeric peptides, and, thus, provide a more specific tumor targeting.
The team connected the multimeric ligand to the anticancer agent cryptophycin‐55 glycinate (Cry-55gly, pictured in red) with a cleavable valine-citrulline (Val-Cit) linker (pictured in green) and a PEG-5 spacer. The resulting multimeric conjugate kills cancer cells expressing integrin αvβ3 more effectively than the analogous monomeric conjugate (5). The multimeric conjugate is non-toxic against control cells which do not express the integrin. According to the researchers, this targeting approach offers increased selectivity in the delivery of drugs.

- Synthesis and Biological Characterization of Monomeric and Tetrameric RGD‐Cryptophycin Conjugates,
Adina Borbély, Fabien Thoreau, Eduard Figueras, Malika Kadri, Jean‐Luc Coll, Didier Boturyn, Norbert Sewald,
Chem. Eur. J. 2020.
https://doi.org/10.1002/chem.201905437