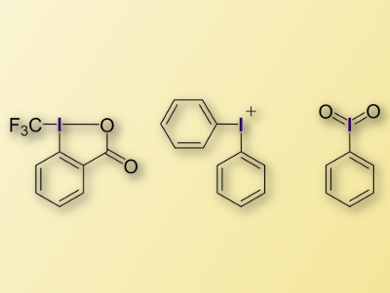
Hypervalent iodane compounds are useful compounds in organic synthesis. Their remarkable reactivity is often associated with their hypervalency, meaning their coordination numbers seem to violate the octet rule. However, phenomena such as three-center bonds or dative bonds can explain such coordination numbers with a maximum of four valence electron pairs.
András Stirling, Research Centre for Natural Sciences, Budapest, Hungary, has performed a computational study to improve the understanding of the bonding situations in such iodanes (examples pictured). The first step was to build a realistic model for the calculation: an iodane molecule was embedded in a large number of solvent molecules (more than 100 molecules), then molecular dynamics (MD) simulations were performed. The electronic structure of the iodane molecule was described very accurately with quantum chemical (QC) methods, whereas the solvent molecules were described approximately (QM/MM simulations).
Using a localization of the electronic orbitals, it was found that the valence of iodine in these compounds is never hypervalent (i.e., larger than four). In some cases, however, it is two or three, which indicates hypovalency. These hypovalent cases can be associated with enhanced reactivity. The results could provide guidance for the design of new iodane molecules.
- Assessing Hypervalency in Iodanes,
András Stirling,
Chem. Eur. J. 2017.
https://doi.org/10.1002/chem.201705285