α,β-Unsaturated carboxylic acids are important substructure motifs widely existing in biologically active natural products and medicines and can serve as useful building blocks in organic synthesis. These compounds are usually constructed by the Wittig or Horner–Wadsworth–Emmons reaction through condensation of aldehydes or ketones with stabilized phosphonium ylides or phosphoryl carbanions bearing a CO2R substituent. However, these reactions always yield a stoichiometric amount of unwanted byproducts with relatively large molecular weights, such as phosphine oxides and phosphate salts, thus limiting their synthetic utility especially in large-scale preparations.
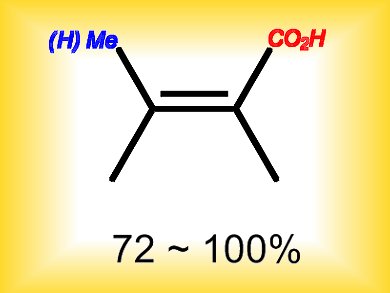
Masanori Takimoto and Zhaomin Hou, Organometallic Chemistry Laboratory, RIKEN, Japan, have reported the efficient preparation of a variety of α,β-unsaturated carboxylic acids, which were difficult to prepare previously, by using a regio- and stereospecific, copper-catalyzed formal methylative and hydrogenative carboxylation of alkynes with CO2.

The use of CO2 as a starting material and simple one-pot nature of this approach should make it a practically useful and attractive method for the synthesis of various α,β-unsaturated carboxylic acids with desired configurations.
- Cu-Catalyzed Formal Methylative and Hydrogenative Carboxylation of Alkynes with Carbon Dioxide: Efficient Synthesis of α,β-Unsaturated Carboxylic Acids,
Masanori Takimoto, Zhaomin Hou,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201301456



![Synthesis of [c2]Daisy Chains via Mechanochemistry](https://www.chemistryviews.org/wp-content/uploads/2025/04/202504_RotaxanesWithSolidStateMechanochemistry-125x94.png)