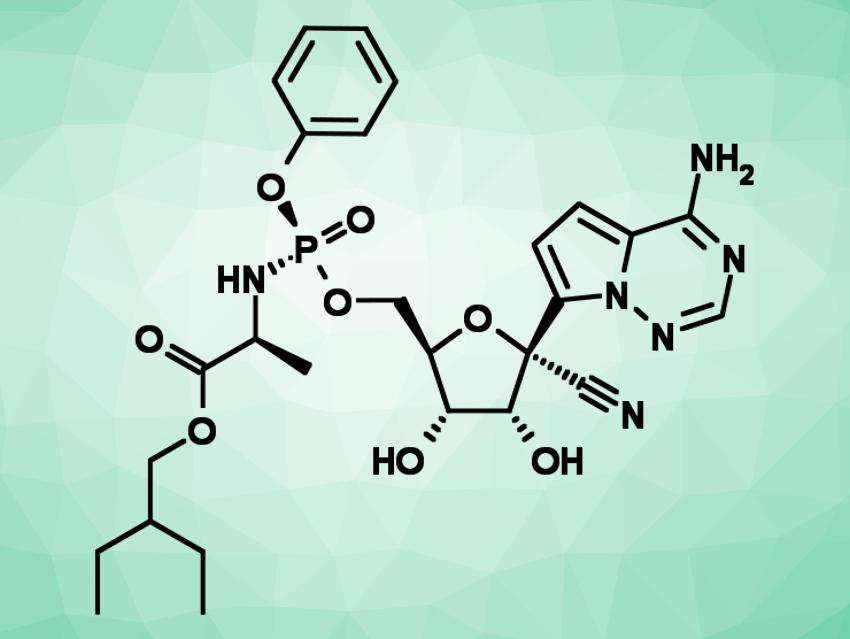
Middle East Respiratory Syndrome (MERS) is caused by a coronavirus and was first reported in 2012. Its high fatality rate means that there is an urgent need for effective antiviral treatments. These treatments could potentially also be useful in the fight against the current coronavirus disease Covid-19.
Emmie de Wit, National Institute of Allergy and Infectious Diseases, National Institute Of Health (NIH), Hamilton, MT, USA, and colleagues have tested whether the antiviral drug remdesivir (pictured) could be used to treat MERS. Remdesivir is a nucleotide prodrug that shows a broad activity against different families of viruses. The team investigated the use of the drug for the prophylaxis and therapy of MERS in a monkey model (rhesus macaques). The monkeys were divided into groups and treated with either remdesivir or a control solution without the active substance, either before or after infection with the MERS coronavirus.
The researchers found that prophylactic treatment with remdesivir prevented the outbreak of the disease. Treatment after the infection reduced the severity of the symptoms, including lung damage, and shortened the disease. The drug inhibits the replication of the virus, which is consistent with previous work showing that remdesivir inhibits the replication of a wide range of coronaviruses.
Due to these promising results and the previous use of remdesivir in humans to treat Ebola, the team suggests testing remdesivir treatment of MERS in a clinical trial. According to the researchers, the work also indicates that the drug could be useful for treating Covid-19.
- Prophylactic and therapeutic remdesivir (GS-5734) treatment in the rhesus macaque model of MERS-CoV infection,
Emmie de Wit, Friederike Feldmann, Jacqueline Cronin, Robert Jordan, Atsushi Okumura, Tina Thomas, Dana Scott, Tomas Cihlar, Heinz Feldmann,
Proc. Natl. Acad. Sci. USA 2020.
https://doi.org/10.1073/pnas.1922083117
Also of Interest
- How Long Can Coronaviruses Live on Surfaces?,
ChemistryViews.org 2020.
Literature review finds coronaviruses could persist on hard surfaces for up to nine days, but can be killed with the right disinfectant - Suggesting Potential Drug Candidates for Coronavirus,
ChemViews Mag. 2020.
Summarizing the current research and suggesting potential drug candidates for treating patients suffering from the 2019-nCoV acute respiratory disease
- LitCovid
Curated literature hub for tracking up-to-date scientific information about COVID-19 - Many publishers and other entities have signed a joint statement to ensure that COVID-19 research findings and data are shared rapidly and openly