Carbonyl compounds are very useful reagents in organic synthesis. They can be synthesized, e.g., by the oxidation of the corresponding alcohols. However, these reactions usually require stochiometric oxidants such as manganese oxide, permanganate, or osmium oxide. Using the oxygen in the air as an oxidant instead would be safer, more environmentally friendly, and cost-effective.
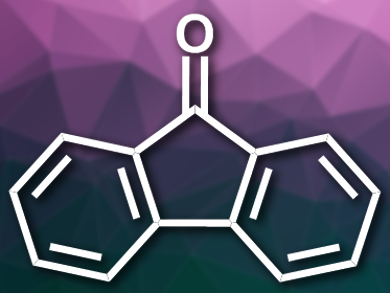
Shoubhik Das, University of Göttingen, Germany, and colleagues have found that 9-fluorenone (pictured) can be used as a metal-free photocatalyst for the light-induced oxidation of alcohols to carbonyl compounds with O2. Using this catalyst, the team oxidized a range of primary and secondary alcohols with dimethyl sulfoxide (DMSO) as a solvent and a blue light-emitting diode (LED) for activation at room temperature in air.
The reaction proceeds in good to excellent yields and has a wide substrate range including aliphatic, alicyclic, and heteroaromatic alcohols. The proposed mechanism includes the excitation of the catalyst by light, a single electron transfer (SET) involving the alcohol, the generation of the reactive oxygen species by the catalyst’s radical anion, and the abstraction of hydrogen from the activated alcohol. The catalytic system was also used for the oxidation of steroids, an important reaction in the production of pharmaceuticals.
- Metal-Free Catalyst for Visible-Light-Induced Oxidation of Unactivated Alcohols Using Air/Oxygen as an Oxidant,
Waldemar Schilling, Daniel Riemer, Yu Zhang, Nareh Hatami, Shoubhik Das,
ACS Catal. 2018.
https://doi.org/10.1021/acscatal.8b01067